Medical Education Metrics (MEMS)
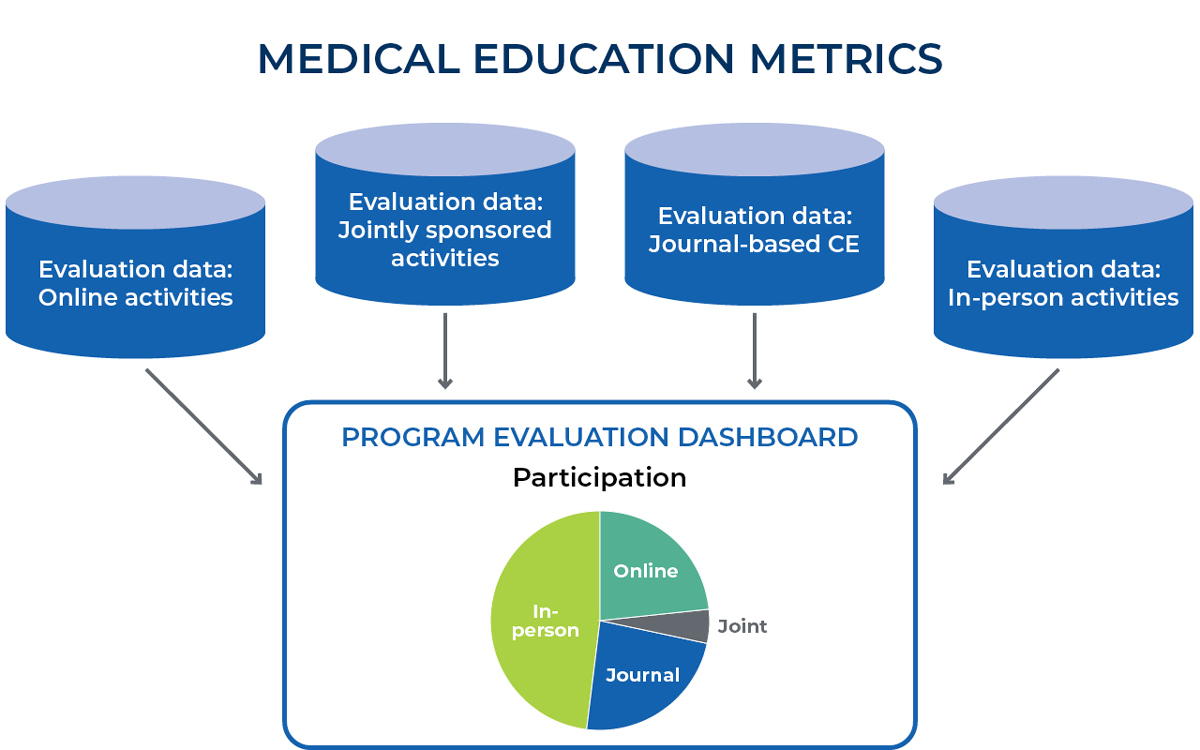
Medical Education Metrics (MEMS) 2.0 enables the exchange of continuing education (CE) data related to the Food and Drug Administration’s (FDA’s) Risk Evaluation and Mitigation Strategy (REMS) for Opioids. Using MEMS 2.0, accreditors collect CE data from providers in a common format, which allows that data to be compiled across accreditation systems for reporting to the FDA. The data allows the FDA to get a sense of how many — and which — clinicians are participating in REMS CE. Data on 839 opioid CE activities and 400,000+ learners has been reported to the FDA so far.
Mission
Develop XML standards and web services requirements and descriptions for the exchange of aggregate evaluation data and other key metrics for health professions education.
Benefits
- Enables collection and analysis of comprehensive CE outcomes data from multiple systems.
- Allows for easier collaboration with partners on the delivery and evaluation of CE.
- Facilitates the development of standardized evaluation instruments.
- Creates opportunities for industrywide data collection and research.
Charter
Download Standards and Guidelines
MedBiquitous standards and guidelines are freely available under an open license. Use the links below to download the standards and guidelines.